 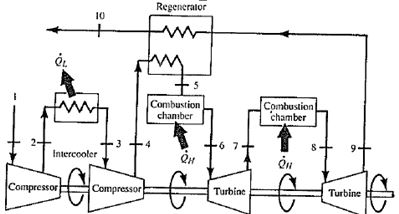
Now, let assume the ideal Brayton cycle that describes the workings of a constant pressure heat engine. Engineers use the specific enthalpy in thermodynamic analysis more than the enthalpy itself. The enthalpy can be made into an intensive, or specific, variable by dividing by the mass. in isentropic process, the enthalpy change equals the flow process work done on or by the system:ĭH = Vdp → W = H 4 – H 3 → H 4 – H 3 = C p (T 4 – T 3 ) For ideal gas αT = 1 and therefore:Īt constant pressure, the enthalpy change equals the energy transferred from the environment through heating:ĭH = dQ → Q = H 3 – H 2 → H 3 – H 2 = C p (T 3 – T 2 )Īt constant entropy, i.e. Where C p is the heat capacity at constant pressure and α is the coefficient of (cubic) thermal expansion. There are expressions in terms of more familiar variables such as temperature and pressure: As can be seen, this form of the law simplifies the description of energy transfer. This work, Vdp, is used for open flow systems like a turbine or a pump in which there is a “dp”, i.e. In this equation the term Vdp is a flow process work. To calculate the thermal efficiency of the Brayton cycle (single compressor and single turbine) engineers use the first law of thermodynamics in terms of enthalpy rather than in terms of internal energy. This is very useful formula, but here we express the thermal efficiency using the first law in terms of enthalpy. Mitsubishi Heavy Industries Technical Review. “Approach to High Efficiency Diesel and Gas Engines” (PDF). Takaishi, Tatsuo Numata, Akira Nakano, Ryouji Sakaguchi, Katsuhiko (March 2008). Therefore we can rewrite the formula for thermal efficiency as: Since energy is conserved according to the first law of thermodynamics and energy cannot be be converted to work completely, the heat input, Q H, must equal the work done, W, plus the heat that must be dissipated as waste heat Q C into the environment. The thermal efficiency, η th, represents the fraction of heat, Q H, that is converted to work. Not included in $W_S$ is the work required to push working fluid into- and out of the device.In general the thermal efficiency, η th, of any heat engine is defined as the ratio of the work it does, W, to the heat input at the high temperature, Q H. $$\dot$ is the mass flow rate through the compressor or turbine, $\Delta h$ is the change in enthalpy per unit mass of the working fluid in passing through the device, and $W_S$ is the so-called "shaft work." This is not the total amount of work, but only the part of the work delivered to- or derived from the rotating shaft. That said, for a compressor or turbine operating adiabatically at steady state, the open system version of the first law of thermodynamics tells us that: For an engineer, understanding the relationship between the closed system version of the first law and the open system version (and how to apply the latter) is vital (in my judgment), particularly when dealing with power cycles such as in your present problem. You said that you want to understand the fundamentals and don't want to memorize stuff. So your first step is to go back to your textbook and get an understanding of this derivation. It requires too extensive a derivation if you are unfamiliar with the open system version of the first law of thermodynamics for a system operating at steady state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
